BioSig Technologies Inc. could be poised to break into the nearly $5 billion global electrophysiology market after nearly a decade of research and development.
The West Los Angeles-based firm filed for federal approval last spring for a medical device that can help doctors target the cells that cause arrhythmias, or irregular heartbeats, which increase the risks of cardiac problems and stroke.
The device could be on the market by this time next year if it passes muster with regulators, the company said.
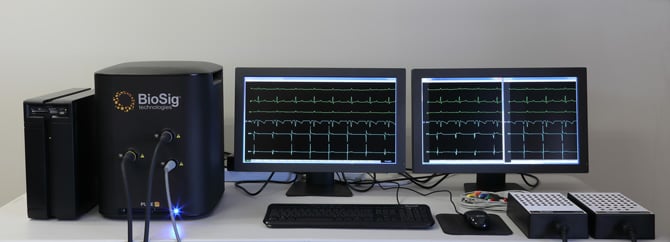
The biomedical device, known as Pure EP, is designed to cut through the background noise of the operating room and its equipment, allowing doctors to pinpoint parts of the heart that cause atrial fibrillation and ventricular tachycardia.
Pure EP displays the heart’s bioelectronic signals on monitors as clearly defined graphs that BioSig says can be seen easily and quickly by doctors, so they don’t have to stop and review the signals for accuracy or filter them.
“What Dolby is to a sound system, we are to the operating room,” said Kenneth Londoner, BioSig’s co-founder, chief executive and investor. “We remove all interference and artifact, and present clear information to the surgeons, to help guide their procedures.”
Atrial fibrillation, the most common arrhythmia, affects nearly 34 million people worldwide, including 6.1 million in the U.S.
The $4.6 billion global market for electrophysiology, which uses catheters to neutralize areas of the heart that can cause irregular heartbeats, is expected to reach $8.3 billion by 2022, according to Allied Market Research.
Precise treatment
BioSig’s device is a biomedical signal processing platform that helps cardiologists perform catheter ablation, a procedure that can lessen arrhythmias.
Pure EP aims to decipher the high-frequency signals that tell doctors which abnormal electrical pathways in the heart have caused the irregular heartbeat. After they “burn” the offending tissue with radiofrequency energy, Pure EP can tell doctors whether they were successful or not. The more accurate targeting can reduce the number of subsequent procedures, BioSig said.
“We’re taking signal processing that’s been around for many years and applying it to electrophysiology,” Londoner said. “We’re giving them the precise information that they never had before.”
BioSig shares trade over the counter. The company is pre-revenue, posted a loss of $12.8 million last year, and has raised $35 million in funding, including $3.5 million from Londoner.
It has 19 employees in Los Angeles, and an administrative office in Austin, Texas.
BioSig has performed 11 pre-clinical studies of Pure EP at Mayo Clinic in Rochester, Minn., Mount Sinai Hospital in New York, Brigham & Women’s Hospital in Boston, and UCLA.
Emerging field
BioSig’s cardiac signal acquisition and display system aims to join an emerging field of bioelectric medicine – which fights disease by targeting electrical signals in the body. Some big names are competing, including GE Healthcare – the health care arm of General Electric Co., Siemens AG, Boston Scientific Corp. and St. Jude Medical Inc., owned by Abbott Laboratories.
It’s been a little more than a year since BioSig signed a 10-year licensing agreement with the commercialization arm of Mayo Clinic for joint development of Pure EP. It has contracted with Minnetronix, of St. Paul, Minn., to manufacture Pure EP for market.
Advisory board
Londoner, BioSig’s co-founder and chief executive, also has extensive corporate experience. He’s founded several health care and logistics companies, a hedge fund that eventually folded, and managed $3.5 billion in assets for J. & W. Seligman & Co., now a subsidiary of Ameriprise Financial Inc.
He shifted his focus to health care and is now a management partner of Endicott Management Partners, a private equity fund in New York focused on medical and consumer-related technology.
The federal clearance for medical devices can take six to 12 months. A best case would allow BioSig’s Pure EP device to launch in 2019, which could open the door for human trials.
“We’re optimistic that, over the next 10 years, we’ll be a force in medicine,” Londoner said, “delivering electricity to the body to treat chronic illness that drugs or surgeries have not optimized or cured.”